Mitochondrial phosphatase PGAM5 regulates Keap1-mediated Bcl-xL degradation and controls cardiomyocyte apoptosis driven by myocardial ischemia/reperfusion injury |
| |
| Authors: | Chen Yang Xiaojuan Liu Fangfang Yang Weixun Zhang Zihao Chen Daliang Yan Qingsheng You Xiang Wu |
| |
| Affiliation: | 1.Department of Thoracic Surgery,Affiliated Hospital of Nantong University,Nantong,China;2.Department of Pathogen Biology, Medical College,Nantong University,Nantong,China;3.Jiangsu Province Key Laboratory for Inflammation and Molecular Drug Target, Medical College,Nantong University,Nantong,China;4.Department of Infection Diseases,Affiliated Hospital of Nantong University,Nantong,China;5.Medical College,Nantong University,Nantong,China;6.Department of Vasculocardiology,Affiliated Hospital of Nantong University,Nantong,China |
| |
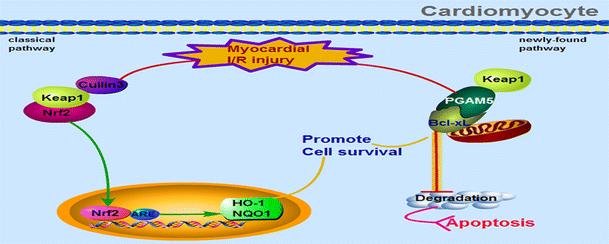
| Abstract: | Phosphoglycerate mutase 5 (PGAM5) is a mitochondrial membrane protein that plays crucial roles in necroptosis and apoptosis. Though PGAM5 is known to be required for inducing intrinsic apoptosis through interacting with BCL2 associated X protein (Bax) and dynamin-related protein 1 (Drp1), the expression and role of PGAM5 in cardiomyocyte apoptosis driven by myocardial ischemia/reperfusion injury(MIRI) has not been studied. The present study shows that PGAM5 expression decreased after MIRI in vivo, positively correlated with Bcl-xL expression, negatively correlated with Kelch-ECH associating protein 1 (Keap1) expression. Furthermore, PGAM5 expression also decreased in cardiomyocytes after hypoxia/reoxygenation (H/R) treatment in vitro. PGAM5 silence promoted cardiomyocyte apoptosis and inhibited Bcl-xL expression, but with no effect on Keap1 expression. Accordingly, Keap1 overexpression further inhibited Bcl-xL and PGAM5 expression. Additionally, PGAM5-Bcl-xL-Keap1 interaction was identified, suggesting that PGAM5 might participate in the degradation of Bcl-xL mediated by Keap1. In summary, PGAM5 controls cardiomyocyte apoptosis induced by MIRI through regulating Keap1-mediated Bcl-xL degradation, which may supply a novel molecular target for acute myocardial infarction (AMI) therapy. |
| |
| Keywords: | |
| 本文献已被 SpringerLink 等数据库收录! |
|