

植物生态学报 ›› 2007, Vol. 31 ›› Issue (3): 490-496.DOI: 10.17521/cjpe.2007.0061
收稿日期:2006-03-24
接受日期:2006-07-31
出版日期:2007-03-24
发布日期:2007-05-30
通讯作者:
许晓明
作者简介:* E-mail: xuxm@njau.edu.cn基金资助:
XIAO Yue-E1, CHEN Kai-Ning2, DAI Xin-Bin1, XU Xiao-Ming1,*( )
)
Received:2006-03-24
Accepted:2006-07-31
Online:2007-03-24
Published:2007-05-30
Contact:
XU Xiao-Ming
摘要:
该文通过pH值漂移实验比较了太湖常见的两种沉水植物菹草(Potamogeton crispus)和马来眼子菜(P. malaianus)对无机碳利用效率的差异,并测定两者无机碳吸收关键酶——碳酸酐酶的活性,探讨了两者无机碳吸收效率差异的原因。根据太湖自然水体的无机碳条件设定了3种不同碱度条件,测定起点pH值和无机碳条件。不同碱度下pH值漂移变化和总无机碳/碱度比值的结果表明,两个种均能利用${HCO_{3}}^{-}$,适应低无机碳条件。两者对${HCO_{3}}^{-}$的吸收速率决定于其浓度大小,该离子浓度越大,光合速率越高。但是对${HCO_{3}}^{-}$的吸收速率存在差异:马来眼子菜在各碱度下终点pH值显著高于菹草,整体光合速率较高。CO2-光合速率响应曲线表明,在高pH值(CO2受到限制)时,马来眼子菜对CO2亲和力较大。尽管菹草在pH值较低(6.5~7.0)时有相对较高的光合速率,但是基于太湖自然水体夏季高pH值(>8.5)条件,马来眼子菜具有更大的生长优势,成为优势种群。两者无机碳吸收速率的差异是造成它们生活史差异和时间生态位的一重要原因。同时,马来眼子菜碳酸酐酶活性明显高于菹草,表明在相同无机碳条件下,前者催化${HCO_{3}}^{-}$与CO2之间的转化效率更高,这可能是造成两者无机碳吸收速率差异的原因。
肖月娥, 陈开宁, 戴新宾, 许晓明. 太湖两种大型沉水植物无机碳利用效率差异及其机理. 植物生态学报, 2007, 31(3): 490-496. DOI: 10.17521/cjpe.2007.0061
XIAO Yue-E, CHEN Kai-Ning, DAI Xin-Bin, XU Xiao-Ming. DISSOLVED INORGANIC CARBON UPTAKE IN TWO SUBMERGED MACROPHYTES FROM TAIHU LAKE, CHINA. Chinese Journal of Plant Ecology, 2007, 31(3): 490-496. DOI: 10.17521/cjpe.2007.0061
| 碱度 Alkalinity (μmol·L-1) | [CT] (mmol·L-1) | [CO2] (μmol·L-1) | 终点pH值 pH values | [CT]/Alk | |
|---|---|---|---|---|---|
| 马来眼子菜 Potamogeton malaianus | 1 200 | 0.832±0.008 | 0.079±0.003 | 10.07±0.01 | 0.693±0.048* |
| 1 600 | 0.933±0.005 | 0.085±0.006 | 10.15±0.05* | 0.583±0.032* | |
| 2 000 | 0.906±0.006 | 0.066±0.003 | 10.22±0.03* | 0.453±0.026 | |
| 菹草 P. malaianus | 1 200 | 0.935±0.007 | 0.210±0.071* | 9.86±0.03 | 0.779±0.034 |
| 1 600 | 0.983±0.005* | 0.127±0.005 | 9.93±0.02 | 0.614±0.005 | |
| 2 000 | 0.938±0.007* | 0.119±0.065 | 10.02±0.04 | 0.469±0.025 |
表1 两种沉水植物在不同碱度条件下终点[CT]、pH值、[CO2]以及[CT]/Alk比值
Table 1 The pH values, [CO2], [CT] and [CT]/Alk quotients reached in pH drift experiments in two submerged macropytes at three alkalinity
| 碱度 Alkalinity (μmol·L-1) | [CT] (mmol·L-1) | [CO2] (μmol·L-1) | 终点pH值 pH values | [CT]/Alk | |
|---|---|---|---|---|---|
| 马来眼子菜 Potamogeton malaianus | 1 200 | 0.832±0.008 | 0.079±0.003 | 10.07±0.01 | 0.693±0.048* |
| 1 600 | 0.933±0.005 | 0.085±0.006 | 10.15±0.05* | 0.583±0.032* | |
| 2 000 | 0.906±0.006 | 0.066±0.003 | 10.22±0.03* | 0.453±0.026 | |
| 菹草 P. malaianus | 1 200 | 0.935±0.007 | 0.210±0.071* | 9.86±0.03 | 0.779±0.034 |
| 1 600 | 0.983±0.005* | 0.127±0.005 | 9.93±0.02 | 0.614±0.005 | |
| 2 000 | 0.938±0.007* | 0.119±0.065 | 10.02±0.04 | 0.469±0.025 |
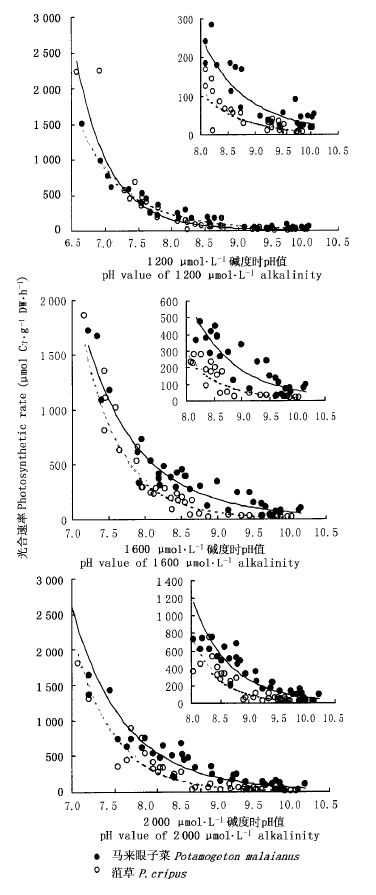
图1 不同碱度条件下两种沉水植物pH值-光合速率响应曲线 实线和虚线分别代表马来眼子菜和菹草的光合速率响应曲线,右上角的各小图为pH值为8.0~10.5的放大图
Fig.1 Photosynthetic rates-pH curves of two submerged macrophytes in three different alkalinities Real lines and dotted lines indicated photosynthetic rates-pH curve of Potamogeton malaianus and P. cripus respectively, and above right figures are all enlarged when pH values were from 8.0 to 10.5
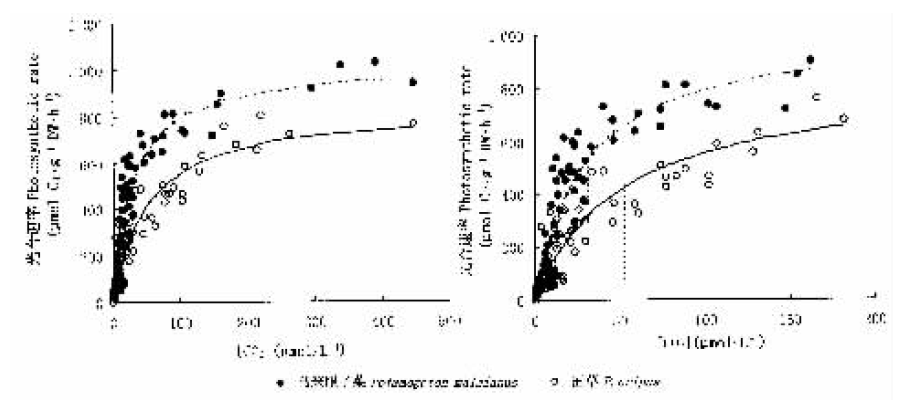
图3 两种沉水植物的CO2-光合速率响应比较 右图为CO2低于200 μmol·L-1时的光合速率响应曲线,R2(马来眼子菜)=0.928,R2(菹草)=0.899
Fig.3 Photosynthetic rates-CO2 curves of two submerged macrophytes Right figure indicated the photosynthetic rates-CO2 curve when the concentration CO2 were lower than 200 μmol·L-1 with R2 (Potamogeton malaianus)=0.928 and R2 (P. cripus)=0.899
| [1] | Allen ED, Spence DHN (1981). The differential ability of aquatic plants to utilize the inorganic carbon supply in fresh waters. New Phytologist, 87, 269-283. |
| [2] |
Badger MR, Price GD (1994). The role of carbonic anhydrase in photosynthesis. Annual Review of Plant Physiology and Plant Molecular Biology, 45, 369-392.
DOI URL |
| [3] | Brownell PF, Bielig LM, Grof CPL (1991). Increased carbonic anhydrase activity in leaves of sodium-defient C4 plants. Australian Journal of Plant Physiology, 18, 589-592. |
| [4] |
Burnell JN, Hatch MD (1990). Low bundle sheath carbonic anhydrase is apparently essential for effective C4 pathway operation. Plant Physiology, 86, 1252-1256.
DOI URL PMID |
| [5] |
Casati P, Lara MV, Andreo CS (2000). Induction of a C4-like mechanism of CO2 fixation in Egeria densa, a submersed aquatic species. Plant Physiology, 123, 1611-1621.
DOI URL PMID |
| [6] | Dai XB (戴新宾) (2001). Effects of carbonic anhydrase in wheat leaf on photosynthetic function under low CO2 concentration. Scientia Agricultura Sinica (中国农业科学), 34, 1-4. (in Chinese with English abstract) |
| [7] | Diao ZS (刁正俗) (1983). The Common Weed of Paddy Field in China (中国常见水田杂草). Chongqing Press, Chongqing, 152-157. (in Chinese) |
| [8] | Jones JI, Eaton JW, Hardwick K (2000). The effect of changing environmental variables in the surrounding water on the physiology of Elodea nuttallii. Aquatic Botany, 66, 115-129. |
| [9] | Kadono Y (1980). Photosynthetic carbon source in some Potamogeton species. Botanical Magazine of Tokoyo, 93, 185-194. |
| [10] | Lampert W, Sommer U (1997). Limnoecology: the Ecology of Lakes and Streams. Oxford University Press, New York, 382. |
| [11] | Maberly SC, Madsen TV (1998). Affinity for CO2 in relation to the ability of freshwater macrophytes to use ${HCO_{3}}^{-}$. Functional Ecology, 12, 99-106. |
| [12] | Maberly SC, Spence DHN (1983). Photosynthetic inorganic carbon use by freshwater plants. Journal of Ecology, 71, 705-724. |
| [13] | Madsen TV (1993). Growth and photosynthetic acclimation by Ranunculus aquatolis L. in response to inorganic carbon availability. New Phytologist, 125, 707-715. |
| [14] |
Madsen TV, Maberly SC (1991). Diurnal variation in light and carbon limitation of photosynthesis by two species of submerged freshwater macrophyte with a differential ability to use bicarbonate. Freshwater Biology, 26, 175-187.
DOI URL |
| [15] | Parsons T, Maita YR, Lalli CM (1989). A Manual of Chemical and Biological Methods for Seawater Analysis. Pergmon Press, London, 142-148. |
| [16] |
Pierini SA, Thomaz SM (2004). Effects of inorganic carbon source on photosynthetic rates of Egeria najas Planchon and Egeria densa Planchon (Hydrocharitaceae). Aquatic Botany, 78, 135-146.
DOI URL |
| [17] |
Portielje R, Lijklema L (1995). Carbon dioxide fluxes across the air-water interface and its impact on carbon availability. Limnology and Oceanography, 40, 690-699.
DOI URL |
| [18] | Sand-Jensen K (1983). Photosynthetic carbon sources of stream macrophytes. Jounal of Experienmntal Botany, 139, 198-210. |
| [19] |
Sand-Jensen K, Gordon DM (1986). Variable ${HCO_{3}}^{-}$ affinity of Elodea canadensis Michaux in response to different ${HCO_{3}}^{-}$ and CO2 concentrations during growth. Oecologia, 70, 426-432.
DOI URL PMID |
| [20] |
Smart RM, Barko JW (1985). Laboratory culture of submersed freshwater macrophytes on natural sediments. Aquatic Botany, 21, 251-263.
DOI URL |
| [21] | Sültemeyer D, Schemidt C, Fock HP (1993). Carbonic anhydrase in higher plants and aquatic microorginisms. Plant Physiology, 88, 179-190. |
| [22] | Vadstrup M, Madsen TM (1995). Growth limitation of submerged aquatic macrophytes by inorganic carbon. Freshwater Biology, 34, 411-419. |
| [23] | Yu D (于丹) (1994). Study on the dynamics and succession of aquatic macrophyte community in the Zhushun Lake, Harbin. Acta Ecologica Sinica (植物生态学报), 18, 372-378. (in Chinese with English abstract) |
| [1] | 冯旭飞, 雷长英, 张玉洁, 向导, 杨明凤, 张旺锋, 张亚黎. 棉花花铃期叶片氮分配对光合氮利用效率的影响[J]. 植物生态学报, 2023, 47(11): 1600-1610. |
| [2] | 林雍, 陈智, 杨萌, 陈世苹, 高艳红, 刘冉, 郝彦宾, 辛晓平, 周莉, 于贵瑞. 中国干旱半干旱区生态系统光合参数的时空变异及其影响因素[J]. 植物生态学报, 2022, 46(12): 1461-1472. |
| [3] | 宋慧清, 倪鸣源, 朱师丹. 乔木与木质藤本的水力与光合性状的差异: 以热带森林崖豆藤属和买麻藤属为例[J]. 植物生态学报, 2020, 44(3): 192-204. |
| [4] | 朱启林, 向蕊, 汤利, 龙光强. 间作对氮调控玉米光合速率和光合氮利用效率的影响[J]. 植物生态学报, 2018, 42(6): 672-680. |
| [5] | 李志民, 王传宽, 罗丹丹. 兴安落叶松叶水力与光合性状的变异性和相关性[J]. 植物生态学报, 2017, 41(11): 1140-1148. |
| [6] | 沈佳, 李亚鹤, 张琳, 孙雪. 蛋白核小球藻生长和无机碳利用对不同光照强度和CO2浓度的响应[J]. 植物生态学报, 2016, 40(9): 933-941. |
| [7] | 赵文赛, 孙永林, 刘西平. 干旱-复水-再干旱处理对玉米光合能力和生长的影响[J]. 植物生态学报, 2016, 40(6): 594-603. |
| [8] | 张蓓蓓, 刘芳, 丁金枝, 房凯, 杨贵彪, 刘莉, 陈永亮, 李飞, 杨元合. 青藏高原高寒草地3米深度土壤无机碳库及分布特征[J]. 植物生态学报, 2016, 40(2): 93-101. |
| [9] | 司晓林, 王文银, 高小刚, 徐当会. 氮硅添加对高寒草甸垂穗披碱草叶片全氮含量及净光合速率的影响[J]. 植物生态学报, 2016, 40(12): 1238-1244. |
| [10] | 唐海萍, 薛海丽, 房飞. 叶片和群落尺度净光合速率关系的探讨[J]. 植物生态学报, 2015, 39(9): 924-931. |
| [11] | 杨浩, 罗亚晨. 糙隐子草功能性状对氮添加和干旱的响应[J]. 植物生态学报, 2015, 39(1): 32-42. |
| [12] | 王荣荣, 夏江宝, 杨吉华, 赵艳云, 刘京涛, 孙景宽. 贝壳砂生境干旱胁迫下杠柳叶片光合光响应模型比较[J]. 植物生态学报, 2013, 37(2): 111-121. |
| [13] | 王建林, 温学发, 赵风华, 房全孝, 杨新民. CO2浓度倍增对8种作物叶片光合作用、蒸腾作用和水分利用效率的影响[J]. 植物生态学报, 2012, 36(5): 438-446. |
| [14] | 陈卫英, 陈真勇, 罗辅燕, 彭正松, 余懋群. 光响应曲线的指数改进模型与常用模型比较[J]. 植物生态学报, 2012, 36(12): 1277-1285. |
| [15] | 孙东宝, 王庆锁. 水分对苜蓿叶片光合特性的影响[J]. 植物生态学报, 2012, 36(1): 72-80. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
Copyright © 2022 版权所有 《植物生态学报》编辑部
地址: 北京香山南辛村20号, 邮编: 100093
Tel.: 010-62836134, 62836138; Fax: 010-82599431; E-mail: apes@ibcas.ac.cn, cjpe@ibcas.ac.cn
备案号: 京ICP备16067583号-19